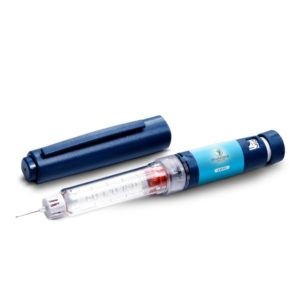
- Home
- Shop
- Peptides
- ACE-031
- AICAR
- AMPK
- AOD-9604
- ARA-290
- B7-33
- BPC-157
- CJC-1295 Dac
- CJC-1295 No Dac
- DSIP
- Epithalon
- Follistatin 344
- FOX04-DRI
- FTPP (Adipotide)
- GDF-8 Myostatin
- GHK-CU
- GHRP-2
- GHRP-6
- GLP-1
- Gonadorelin
- HCG
- Hexarelin
- HGH Fragment 176-191
- HGH191AA
- HMG
- Human C
- Humanin
- IGF-1 DES
- IGF-1 LR3
- Ipamorelin
- Kisspeptin
- KPV
- LL-37 Cap18
- Melanotan 1
- Melanotan 2
- MGF
- MOTS-C
- NAD+
- Oxytocin
- P-21
- PEG-MGF
- PNC 27
- PT-141
- PTD-DBM
- Selank
- Semax
- Sermorelin
- Snap-8
- SS-31 Elamipretide
- TB500
- Tesamorelin
- Tesofensine
- Thymalin
- Thymosin Alpha 1
- Tirzepatide
- Triptorelin
- VIP
- Vitamin B12
- Sarms
- Blended
- Help